Last Update: July 10, 2015


When a crude oil spill disaster strikes there's serious need to contain the oil, and prevent it from harming people or animals or plant-life in the area. One method is to spray dispersants onto the crude oil, in the hope this will limit the damage. They are supposed to break crude oil down into small droplets that then disperse throughout the water, making it more biodegradable by microbes. But that means highly toxic oil, mixed with highly toxic dispersants, are then more thoroughly mixed with seawater, enter the food chain, and the results are little studied or understood.
Crude oil is highly toxic and is full of harmful carcinogenic chemicals. Of course we want a magic wand to wave when crude oil is spilled in an accident, to render the oil harmless. To that end, the oil companies developed oil dispersants.
The problem is that the dispersants in use are themselves extremely toxic, and the use of dispersants makes the oil spill problem very much worse.
An oil dispersant is a mixture of surfactants and solvents that helps break oil into small droplets following an oil spill. The idea is to break down oil droplets to smaller droplets, so the oil mixes with water more readily, doesn't float on the surface, and therefore (theoretically) won't impact the shoreline. Using dispersants to treat an oil spill is a trade-off between the impact on sub-surface marine life, the impact on surface marine life, and the coastal destruction which would occur if spilled crude oil reaches the shoreline. [Wikipedia1]
The crude oil becomes more bio-available to marine life - meaning that more of the crude oil will be injested by marine life. The oil can sink to the ocean depths where the "out of sight out of mind" phenemona makes it look like "problem solved" but in actuality the situation is made far worse.
History of how Dispersants came to be used
One of the first uses of dispersants to mitigate an oil spill came after the wreck of the SS Torrey Canyon on March 18, 1967, after the ship struck Pollard's Rock on Seven Stones reef between the Cornish mainland and the Isles of Scilly. This was a large oil spill which was badly handled, and it ended up affecting hundreds of miles of Cornish, English and French coastline. [Wikipedia2]
At the time the phrase "detergents" was used, probably to reduce the negative perception by using a common word. The compounds were probably variants of products originally formulated to clean surfaces in ships' engine-rooms. No thought was given to the toxicity - in their desperation to clean up the Torrey Canyon oil spill the government made multiple bombing runs on oil slicks to set them on fire. In some cases crude oil that had been treated by dispersants was plowed into beach sand to bury the oil.

At the Exxon Valdez oil spill in Alaska on March 24, 1989, some Corexit was used. A document had been written in 1986, "Oil Dispersant Guidelines for Alaska", saying dispersants were to be used for oil spills that endangered human life, or to prevent or reduce hazard to human life. Another guidance document specifically covering Prince William Sound (the location of the Exxon Valdez incident) was coincidentally published three weeks before the accident. The accident occurred in an area that document labeled "Zone 2", which was recognized as being biologically sensitive and therefore requiring more stringent approvals before using dispersant.
Almost immediately after the spill, Alyeska Pipeline requested permission to use Corexit 9527 dispersants, but were only allowed a dispersant "test" once the leading edge reached another zone that was less biologically sensitive. That test was inconclusive. Exxon took over responsibility shortly later and ran several test applications with Corexit 9527 dispersants, all of which were inclusive. Exxon is reported to have always claimed Corexit gave good results, however. Government representatives became so skeptical, that they completely denied approval for any dispersant use in areas closer to shore. That allowed a few Corexit tests further offshore, all of which were inconclusive. " [PrinceWilliamsSound]
According to
Wikipedia, a 1993 report by the Alaska Department of Environmental Conservation said about dispersant use that "It had not been tested, scientific data on its toxicity were either thin or incomplete, and it had operational problems. In addition, public acceptance of a new, widespread chemical treatment was lacking. To landowners, fishing groups, and conservation organizations, the idea of dumping chemicals on hundreds of miles of shorelines that had just been oiled seemed much too risky - especially when there were other alternatives." It also cites 2-butoxyethanol, a primary component of the Corexit formulation used, as the primary agent causing liver, kidney, lung, nervous system, and blood disorders among cleanup crews in Alaska. [TakePart]

At the
Deepwater Horizon oil disaster in April 2010, over 1.84 million gallons of Corexit was sprayed. [NationalCommissionDeepwaterHorizon] Dispersant use (Corexit) was begun almost immediately after the explosion, because existing contingency plans allowed for dispersant use and Corexit was on the approved list. Over 14,000 gallons of Corexit was applied during the week of April 20-26, 2010. On April 29, 2010, the spill was declared a "Spill of National Significance" and the Commandant of the Coast Guard, Admiral Thad Allen, became the National Incident Commander. Dispersent use increased rapidly after that point in time.
On May 25, 2010, tests for effectiveness and toxicity of subsea dispersants (as opposed to the sort you spray from an airplane) had been completed. The EPA and Coast Guard jointly announced approval for their use.
On May 20, 2010, rising concern about potential toxicity of the dispersants being used caused the Coast Guard and EPA to issue a directive requiring BP to identify and use a less toxic dispersant. They'd primarily used Corexit 9500, and according to data in the National Contingency Plan some of the approved dispersants were both less toxic and more effective than Corexit 9500. BP responded saying that there were only five suitable dispersants on the approved list, and that Corexit 9500A was the most suitable of the bunch. Some other dispersants were known to break down into nonylphenol (a known endocrine disruptor), and complained that manufacturers of other dispersants were keeping the formula's secret. In the case of Corexit 9500, the manufacturer claimed it rapidly broke down within a month in the environment meaning Corexit has fewer long-term side effects than other dispersants.
On May 24, 2010, EPA Administrator Lisa Jackson held a press conference on the issue. She instructed BP to scale back dispersant use, expressed dissatisfaction over BP's efforts to analyze other dispersants, and announced the EPA would conduct their own tests to verify BP's claims about various dispersants. Over the next couple months BP did cut back on dispersant use.
The well was capped in mid-July 2010, by which time dispersant use had stopped completely.
The National Commission Deepwater Horizon report states that the EPA failed to
- consider the possibility that dispersants would be used in the massive volumes required by the Deepwater Horizon spill
- nor consider the possibility that subsea dispersants would be required in such volumes
In May/June a panel of scientists were quickly convened to consider the tradeoff's of massive dispersant use in this disaster. Their answer came on June 4, 2010 -- reporting a consensus that "use of dispersants and the effects of dispersing oil into the water column has generally been less environmentally harmful than allowing the oil to migrate on the surface into the sensitive wetlands and near shore coastal habitats."
It may not be entirely clear from the summary above, but the bottom line in the National Commission Deepwater Horizon report is that very little is understood about the impact of dispersants when applied to spilled crude oil in the environment. The people in charge were blindly making decisions with insufficient data to make correct well reasoned calculations. The report called for more studies of dispersants, and development of "green dispersants".
Dispersant Theory
Dispersants function like detergents, with the goal of breaking oil into small droplets which easily mix with water. It's done with surfactants and solvents. Surfactants are a compound of lipophilic and hydrophilic substances, the result being to reduce surface tension on oil droplets. The solvents help the surfactants pass through the water, and reach the oil-water boundary.
The result is not to render the crude oil harmless, but to mix it with water. The oil becomes suspended in the water rather than rising to the surface.
The operative phrase is "out of sight out of mind". Oil suspended in water isn't visible on the water surface, and therefore stays out of news reports.
The National Commission Deepwater Horizon report linked in the footnotes claim these benefits from using dispersants:
- less oil will float ashore to adversely affect shorelines and fragile estuarine environments.
- animals and birds that float on or wade through the water surface may be less exposed to oil
- dispersants may accelerate the rate at which oil biodegrades. Smaller droplets have a larger surface-area-to-volume ratio, which in theory should allow microorganisms greater access to the oil, and speed their rate of consumption. The expected acceleration of this biodegradation is often cited as a major reason to use dispersants.
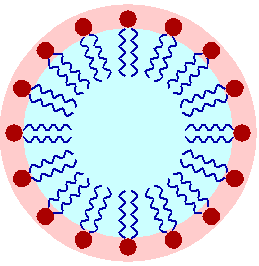
The image to the right shows a schematic diagram of a micelle of oil in aqueous suspension, such as might occur in an emulsion of oil in water. In this example the surfactant molecules' oil-soluble tails project into the oil, while the water-soluble ends remain in contact with the water phase). It's the job of Dispersants to disrupt such oil droplets.
Dispersant Constituents - Surfactants
Surfactants are widely used in industry, and are amphiphilic compounds meaning they contain both hydrophobic groups (flees from water) and hydrophilic groups (bonds with water). That means surfactants contain both water-soluble and water-insoluble components. [Wikipedia3]Surfactants form Micelles such as shown here. The action occurs when the Surfactant Micelle touches an oil Micelle, and the surfactant chemicals cause chemical changes to occur in the oil Micelle so it breaks apart.
A particular Dispersant product will contain multiple chemicals, and each formulation follows different strategies of action. For example the Wikipedia page lists the components of two products (NOTE: Many of the Dispersants have proprietary formulations)
Omni-Clean OSD:
- Sodium lauryl sulfate - Surfactant - Charged ionic surfactant and thickener
- Cocamidopropyl betaine - Surfactant - Emulsifying agent
- Ethoxylated nonylphenol - Surfactant - Petroleum emulsifier & wetting agent
- Lauric acid diethanolamide - Dispersant - Non-ionic viscosity booster & emulsifier
- Diethanolamine - Detergent Water-soluble detergent for cutting oil
- Propylene glycol - Emulsifier - Solvent for oils, wetting agent, emulsifier
- Water - Solvent - Reduces viscosity
- Oleic acid sorbitan monoester - Emulsifying agent
- Coconut oil monoethanolamide - Surfactant - Dissolves oil and water into each other
- Poly(ethylene glycol) monooleate - Surfactant - Oil-soluble surfactant
- Polyethoxylated tallow amine - Surfactant - Oil-soluble surfactant
- Polyethoxylated linear secondary alcohol - Surfactant - Oil-soluble surfactant
- Dipropylene glycol methyl ether - Solvent - Enhances solubility of surfactants in water and oil.
- Water - Solvent - Reduces viscosity

Effects of using dispersants on oil spills
Deepwater Horizon Oil May Sit On Ocean Floor, But How Did It Get There? [NPR1] Oil is supposed to float on the surface (it's lighter than water) so it's surprising to learn there's a layer of oil, on the ocean floor, surrounding the Deepwater Horizon site. A study by a team of scientists looked at oil on the sea-floor surrounding the site, and chemical analysis shows a chemical signature matching oil from the accident. [NationalAcademyofSciences] The researchers looked at coral colonies, most of which had been affected (killed) by exposure to the combination of crude oil and dispersants.
Of course, the oil was on the ocean floor, rather than the surface, because of those dispersants.
Using Natural Abundance Radiocarbon To Trace the Flux of Petrocarbon to the Seafloor Following the Deepwater Horizon Oil Spill [ACS1] A similar study used radiocarbon techniques to map the amount of carbon material on the ocean floor. Because the crude oil doesn't have radioactive carbon 14 it stood out like a sore thumb. Researchers were able to estimate the total amount which settled on the ocean floor in the range of 6-10 million gallons. They warn that this material will become part of the food chain, poisoning fish in that area for decades to come. [PhysOrg1]
"
Oil and oil dispersant do not cause synergistic toxicity to fish embryos." Julie Adams, Michael Sweezey, Peter V. Hodson -- Environmental Toxicology and Chemistry Article first published online: 5 NOV 2013 DOI: 10.1002/etc.2397
This study tried to gauge the relative toxicity of leaving an oil spill alone, versus treating it with a dispersant. The study found that the toxicity of oil in the environment is attributed solely to the oil, and not to synergistic interaction between oil and dispersant. While the dispersants don't increase the toxicity of the oil, they "increase the amount of oil mixed into water by 100-fold compared to undispersed oil floating on the surface" according to Dr. Hodson. He recommended that "The use of dispersants should be restricted to brief applications in open waters where marine life is less abundant." [PhysOrg2]
Kuhl, A. J., Nyman, J. A., Kaller, M. D. and Green, C. C. (2013),
Dispersant and salinity effects on weathering and acute toxicity of South Louisiana crude oil. Environmental Toxicology and Chemistry, 32: 2611–2620. doi: 10.1002/etc.2346
This study tried to measure the toxicity of dispersants and crude oil with biodegradation occuring in water of varying salinity. In other words, what happens to the toxicity of crude oil in the ocean if dispersants are sprayed?
It found that COREXIT use exponentially increased crude oil polycyclic aromatic hydrocarbons (PAHs) concentrations for up to 16 weeks (the total length of the study). Results suggested reduced biodegradation activity in low salinity water.
“Response of Deep-Water Corals to Oil and Chemical Dispersant Exposure” Scientists at Temple Univ studying the Deepwater Horizon aftermath found the COREXIT and Crude Oil combination was lethal to sea coral populations. [TempleEDU] They found several corals covered with slime that came from the oil spill, and was mixed with dispersants. They found that dispersants are more lethal to corals than was the crude oil itself, and that the combination of oil and dispersant increases oil's toxicity.
Synergistic toxicity of Macondo crude oil and dispersant Corexit 9500A® to the Brachionus plicatilis species complex (Rotifera) This study found that this combination (Corexit 9500 and crude oil) is 52x more toxic to Brachionus manjavacas, but that a different species (Brachionus plicatilis) is not sensitive to this combination. [ScienceDaily]
Oil Spill Truth: This blog has a page discussing dispersants and dispersed oil - "
Dispersants and Dispersed Oil". It's claimed that BP Executives and Board Members have direct financial ties with Nalco, the manufacturer of Corexit. During the Deepwater Horizon disaster response, BP had been ordered to completely cease Corexit use but BP ignored the order. The question is raised whether BP did this for sound technical reasons, or because their executives stand to gain financially the more Corexit is used? The same page goes on to note that Corexit is both extremely toxic to certain fish species, and less effective than other dispersants (that are less toxic).
Biodegradability of Corexit 9500 and Dispersed South Louisiana Crude Oil at 5 and 25 °C found that one of the components of Corexit 9500, dioctyl sodium sulfosuccinate (DOSS), degrades much more slowly in cold water than in warm water. The Corexit applied at the Gulf surface degraded as expected, but the Corexit applied below the surface degraded slowly. The results raise concerns not just for deep sea treatment of oil spills, but of spills occurring in the Arctic - an area where the oil companies want to expand operations. [ACS2]
Link between oil spill exposure and hematologic, hepatic toxicity This study looked at health records of oil spill cleanup workers who'd worked on the Deepwater Horizon disaster, and other people who did not do such work. The oil spill cleanup workers had blood results showing severe chemical contamination, with platelet counts drastically decreased and other effects suggesting they'll develop blood related diseases.
Corexit: Deadly Dispersant in Oil Spill Cleanup An updated report published on the fifth anniversary of the Deepwater Horizon disaster, discusses evidence submitted by "whistle blowers" who live and work along the Gulf Coast. It was an update to their 2013 report, Deadly Dispersants in the Gulf: Are Public Health and Environmental Tragedies the New Norm for Oil Spill Cleanups? They documented findings including the following:
- Severe health problems including blood in urine, heart palpitations, kidney damage, migraines, multiple chemical sensitifity, neurological damage, rapid weight loss, respiratory system damage, seizures
- Blood test results showing alarmingly high levels of chemical exposure
- BP and the Government both misrepresented known risks by claiming Corexit is low in toxicity
- While the Occupational Safety and Health Administration (OSHA) developed a highly-regarded safety training program for cleanup workers, the workers did not receive any training
- Federally required worker resource manuals listing Corexit health hazards either were not delivered or were removed from BP work sites
- While government regulations prohibited divers from operating during the spill, divers contracted by the NOAA did so after being told it was safe
- BP was allowed to retaliate against workers who insisted on wearing respirators while working.
- Workers were provided with minimal or no personal protective equipment on the job
Source:
gulfoilspill
Source:
jim bo
Source:
jim bo


